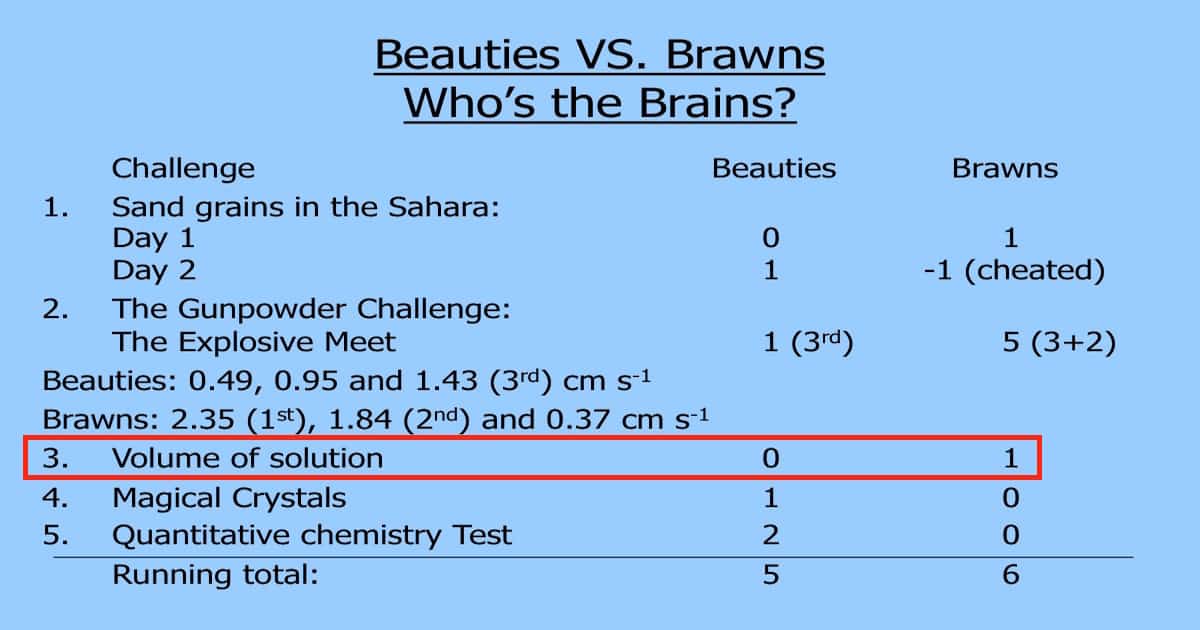
This third challenge for my 2009-10 first year IB Chemistry student in Baku, Azerbaijan was a pretty straight forward one.
All I did was mix equal volumes of two liquids — water and ethanol — in a volumetric flask. (I used a volumetric flask because I found it work best owing to its narrow neck and marking.) All that the Beauties and Brawns had to do was predict whether the volume would add up or would be more than the sum or less than the sum of the two.
The volume actually don’t add up; the final volume comes out to be slightly less. Here’s a clip from an episode of Bill Nye the Science Guy in which they perform the same demonstration and also explain why that happens.
https://youtu.be/84k206qaVRU
If you are a teacher, here’s a number of additional tasks you can set your students.
The explanation is that water molecules fit in space between the alcohol molecules. You can have the student actually do a little bit of research and calculation to see if indeed that is possible.
To begin with, which of the two has bigger space between their molecules? And by how much?
Can one fit in the space of the other? If so, how many? Describe/account for the interaction in terms of their intermolecular forces/bonds.
What would be the result if ethanol were replaced by methanol in the above demonstration? Confirm your hypothesis with calculations similar to the one conducted for ethanol.
In order to figure that out, just as with the sand grains in the Sahara desert challenge, they have to find data or calculate them to help answer this question, and in the process make a number of assumptions as well. Obviously, there is more than one way of getting to the answer just as with many open-ended problems.
Incidentally, the Brawns won a point in this one, as you can also see from the image at the top.
Have you done anything similar in your class? How did it go? Feel free to share below.